Upgrading from Overweight: The Effects of GLP-1 Drugs on the Investment Landscape
Are drugs like Ozempic just a fad or the answer to a problem that the Developed world has been seeking for decades?
Upgrading from Overweight:
The Effects of GLP-1 Drugs on the Investment Landscape
The Questions: GLP-1 Drugs and Obesity
If you haven’t, please read my article on Thematic Investing & Megatrends here first.
“Ozempic” is now, essentially, a byword for “weight loss drug”. It and similar drugs are dominating social media trends to the point of being mentioned at the Oscars, it has the makings of a cultural touchstone. Perhaps, even, a triumph of modern science over a modern disease that has plagued developed countries for decades. I have been long LLY and NVO because of GLP-1 drugs since early 2022, but as evidence continues to amass in favour of their utility for a large population it’s worth considering - what other implications does this have? It is not an exaggeration to say entire industries have been built on the back of the obesity epidemic, its drivers and its consequences. So, the questions remain….
Do these drugs work?
What are the impacts on the wider economy of a drug that is nearly as effective as bariatric surgery and acts via, primarily, appetite reduction?
Which companies will benefit from those trends, besides the ones producing the drugs?
Which companies and industries will suffer?
Are those industries at risk right now and, if not, when?
What level of pragmatism is called for in tempering expectations?
This isn’t just about a drug that works, it’s about a drug that works for something that has reached the status of epidemic in most developed markets. The CDC estimates that nearly 42% of the U.S. population is obese, and most view it in as a moral failing that will never be cured by science. I don’t know the answer to the first part, but the second part seems unlikely.
As an investor I am keen to be educated on what has the makings of a plausible solution. if you’re out of hand dismissing something that can result in 20-25% weight loss in a year, in a country where about a third of the population is obese and your profession is “projecting trends in how consumers and businesses spend and earn money” well…maybe don’t do that.
Already, the impact is being felt, as Walmart attested in their latest earnings call:
This article will hopefully serve as a primer for not just GLP-1’s, but a springboard for a discussion on potential secondary knock-on effects in both patients taking them and the wider economy. Additionally, background, some current arguments around these drugs and their effects, some information in general on peptide medicines is provided (due to various behavioral oddities instilled in me during my short time at medical school, this article will have legitimate academic sources) and, of course, there is a basket of stocks which I believe are either positively impacted, negatively impacted or should simply be mentioned.
Is it finally time to bet against the American Eater?
Let’s find out…
But first, consider subscribing. Paid subscribers received the below on July 11th 2023.1
Introduction
View & Download a spreadsheet of the Thematic Equity Basket, including tickers & weightings, which is discussed in this article here
There are 5 FDA approved drugs for weight loss, currently: orlistat, phentermine-topiramate, naltrexone-bupropion, liraglutide, and semaglutide. A sixth, tirzepatide, is expected to be approved soon. Currently, Saxenda (Liraglutide - NVO) and Wegovy (Semaglutide - NVO) are the two GLP-1 drugs approved for weight loss by the FDA. Both drugs are delivered via subcutaneous (beneath the skin) injection using autoinjectors (patients can self-administer the injection at home). While oral preparations are in development, currently the subcutaneous autoinjectors are the primary delivery method - a boon for companies like Stevanato (STVN) which has managed to take market share from Beckton Dickinson (BDX) in the area.
Saxenda injections are taken once daily and Wegovy once weekly. Eli Lilly, the maker of the drug Mounjaro (Tirzepatide - LLY), said it expects the medication could be approved by the U.S. Food and Drug Administration (FDA) for weight loss as early as the end of the year 1. The company made this prediction on the timing of FDA approval as it announced top-line results of a Phase 3 study showing a nearly 16% overall weight loss over a 17-month period among people who have both Type 2 diabetes and obesity 1. The FDA has not said when it would announce a decision about weight loss approval for Mounjaro, but consensus expectations place it sometime in Q4 23 = Q1 24.
Mounjaro injections are also taken once weekly and, despite only currently being approved by the FDA for diabetes, have shown the most impressive weight loss numbers of any GLP1 drug approved by the FDA in general. Recent data from LLY on Retatrutide (as well as other promising solutions presented at the American Diabetes Association conference) have made me consider something that seems inevitable on the surface, but the timeline of which may be greatly accelerated: obesity may have a cure (specifically - “diabesity”, the word for diabetes and obesity as comorbidities) in these new medications. Regardless of whether this very optimistic statement is true, what is true is that these drugs are effective for most patients who are obese in causing significant BMI reductions that previously would only be achievable with gastric bypass or sleeve gastrectomy.
Misconceptions & Assumptions
1) The biggest “gotcha” on social media surrounding these drugs seems to be some sort of vague speculation that they will melt your insides or otherwise have some horrific side effects a decade later. While there are certainly side effects to these drugs, which we will expand on later, GLP-1 drugs have been on the market for almost two decades. The first GLP-1 (glucagon-like peptide-1) agonist drug approved by the FDA was exenatide. It was marketed under the brand name Byetta and got approval in 2005. GLP-1 drugs are hardly some experimental medicine.
a) Trulicity was approved in 2014 and has been a significantly popular treatment for T2D since. Consistent with other GLP1R agonists, nausea, diarrhea and vomiting are common side-effects, with no long-term harms noted from Trulicity over its 9 year use in the markets. To say that. when it comes to weight loss, these drugs have improved since then is a bit of an understatement:
2) GLP-1 drugs do not “allow you to eat whatever you want”. In fact, it’s quite the opposite. Eli Lily has been one of my largest single stock long positions since 2021 and I have, as you do with any large position, built up a number of channel checks (that is to say, operators and those exposed first hand to the businesses). That includes endocrinologists. Anecdotally, I have heard him say that he has to go as far as recommending protein supplementation2 to patients who are clearly not eating more than 500 calories a day, simply because they do not have the appetite to.3 Patients who, before they were on these drugs, consumed upwards of 5000 calories a day. This is a significant concern for businesses who might be adversely affected by lower volumes of food sales to obese individuals. Not because they won’t eat fast food or junk food anymore, but simply because they will not eat as much of it.
3) Some believe these drugs will not see widespread adoption because they are most effective subcutaneously (administered via an autoinjector as pictured here): While a certain subset will always have a phobia of needles, it’s worth considering the lengths that people have gone to in the past to lose weight (taking pills with tapeworms in them, for example). BofA polls show that 70% of patients are comfortable with the autoinjector. Subjectively I believe there is a significant difference when it comes to a fear of needles when it’s a syringe vs. an autoinjector. Think of an epiPen - you don’t actually see the needle. I think this is an overblown concern
.
4) While it’s probably not a “miracle drug”, it’s not unreasonable to look out in a decade or so and see that we have solved the problem of obesity. Will there still be unhealthy people, with unhealthy habits? Absolutely. But the level of obesity in the US is absolutely a public health crisis, presents massive costs on both a personal and societal level and in large part seems to perpetuate as a disease state (Metabolic Syndrome etc) - it’s completely reasonable to think that modern science can cure this condition which is largely a symptom of modern lifestyles anyway. I think it overly pessimistic to view it as impossible and as an investor believe it may represent a significant shift in the preferences, behaviors and consumption habits of a large portion of the population.
On Wegovy, patients lost an average of 17.4% of their body weight over the 68 week trial. It is even more significant in Mounjaro (tirzepatide) which will likely be FDA approved by the end of the year for weight loss.
Will patients keep the weight off if they go off the drug? So far, with wegovy, patients gain back about half of the weight they lose. In early 2024, we’ll find out how Mounjaro stacks up against this. I think it’s important to note, however, that this does not invalidate the initial weight loss or the potential popularity of these drugs - nor the chances that patients may stay on them indefinitely. It’s worth considering: will patients with Erectile Dysfunction be able to get erect if they don’t take viagra (or another PDE5 Inhibitor)? Will patients with hypothyroidism be able to function normally if they stop taking Levothyroxine? Of course not. That doesn’t change much when it comes to the net effects.
Commentary on Wegovy and expectations for the SURMOUNT Mounjaro trial from LLY management has been:
“Once they stop the medicine. And what you'll see here is the rate of weight regain is about half the rate of weight loss...That's what I would expect. I would expect it's more similar than different to this graph.”
5) There have been significant supply chain issues with all GLP-1 drugs as supply has not kept up with demand. I outline a way to play the “picks and shovels” of this issue, but it is also indicative of the fact that there is significant demand and further impact to be had for this theme - simply put, not enough people are on it yet. But they will be, if history is any indication.
Affordability
Ozempic is very expensive. Will it be less expensive / why will GLP-1s have a significant second order impact worth paying attention to if it does not span across socioeconomic backgrounds to reach the largest demographic of obesity? This is a valid concern, and it is indeed possible that these drugs stay expensive for the next couple years due Eli Lilly and Novo Nordisk’s duopoly, but I think to underestimate the impact they will have because of any delay is short sighted. Even if, in the first couple years of Mounjaro and Wegovy both being approved for the weight loss indication, the impact from just those taking it who can afford it/have the insurance for it can be significant. Over time, factors will conspire to make it more widely covered and cheaper.
Consider this: Viagra, also known by its generic name sildenafil, was approved by the FDA in 1998. At the time of its launch, the average cost of Viagra was approximately $88 per pill. Over the last two decades, sildenafil experienced a drastic price reduction from a peak of $88.45 per pill for the branded version (Viagra 100 mg),11 , 12 to $1-$4 a pill for a generic version. Five or six years after it was first released, it was a drug that was certainly not cost prohibitive for anyone with insurance. Now, even without insurance, most people in the US with ED can afford a prescription. Affordability for drugs in markets with significant demand is typically accelerated. Indeed, if tirzepatide and other GLP-1 drugs for weight loss become subject to mandatory discounting through the Inflation Reduction Act (by 2031, or sooner if their developers attempt to reduce prices in anticipation of this deadline) or similar regulation, their prices would be directly reduced, making them more affordable. Beyond this, there are several other factors that could contribute to increased affordability over the next 5 years:
Competition: Other companies like Viking Therapeutics and Altimmune are developing their own GLP-1 drugs. An increased number of available drugs in this class could drive prices down due to competition.
Insurance Coverage: If tirzepatide is approved by the FDA for weight loss in 2023, insurance companies are more likely to cover it, which would reduce out-of-pocket costs for patients. As more and more data becomes available, the cost benefit analysis of these drugs will become impossible to ignore for health insurance companies.
Generic Drugs: While it could take more than 5 years, eventually patents on GLP-1 drugs will expire, leading to the introduction of generic versions. Generics are typically sold at a fraction of the cost of brand-name drugs. Liraglutide’s patent expires this year in the US and Europe,
Price Negotiation: With more options available in the market, insurers and pharmacy benefits managers may be able to negotiate lower prices with drug manufacturers.
Economy of Scale: As these drugs become more widely used via more delivery mechanisms, manufacturers may achieve economies of scale, reducing production costs and potentially lowering prices.
Right now, Novo has a monopoly on GLP-1 drugs that are FDA approved for weight loss. Although, anecdotally, it is no secret that Mounjaro is already being prescribed to patients without T2D for weight loss and the comorbidity of T2D and obesity blurs the line, once Mounjaro is approved I believe it will begin reducing the average out of pocket costs for these drugs. Lilly’s approval for Mounjaro in Obesity will result in a duopoly in the space, with clinical stage biopharma (mentioned below) likely 2 years from having a drug on the market. Mounjaro has butted in a bit with coupon programs, but still only was approved by health insurers 50% of the time for off label indications. This dynamic makes it simple to own NVO & LLY for the initial duopoly while owning smaller concentrations of the clinical stage companies for the option value once the duopoly is busted.
Still, even as they become more affordable, I believe there are plenty of catalysts in this area to sufficiently propel their manufacturers revenues. If tirzepatide gets approved for additional indications like NASH, sleep apnea, CKD, heart failure, pre-diabetes, etc., it could significantly increase the market opportunity for Eli Lilly. These are all large market diseases with high unmet needs. Particularly, NASH and CKD are areas where successful drug development has been challenging, and a new effective treatment could be a significant revenue driver. Successful drugs often come with high margins, and the successful development and commercialization of tirzepatide and other drugs could drive margin expansion. Further, Eli Lilly could potentially see operating leverage as higher sales spread out the fixed costs of their operations, similar to what occurred with Viagra and Pfizer. Oral GLP-1 options can greatly improve patient compliance and expand the total addressable market. Retatrutide also represents a new class of drugs that could have broad applications, further expanding Eli Lilly's market opportunity.
The endocrinologist I have spoken with regularly about this has mentioned that semaglutide is more muscle sparing than Mounjaro, which raises interesting points about how wide this market may be in terms of each modality perhaps retaining a market share for a very specific type of body composition requirement. Here’s commentary from Lily management:
“There's no doubt in my mind, Geoff in five years, as I said, we're going to have a lot more data and we're going to see these drugs as foundational medical treatment for many chronic diseases, and with outcomes to hard outcomes data that moves the cost curve in populations.”
“And eventually the Venn diagram of seniors who have obesity or overweight and other conditions, diabetes, of course already we have access in many plans there, but we'll be adding to that pie. And then the question of is there straight chronic weight management in the elderly becomes less important over time?”
“And so, this [reimbursement] will play out over time on both as we look at data and outcomes to drive further expansion of access.”
I think the affordability aspect is simply a matter of time. Whether through insurance coverage or competition or both, these drugs solve a problem too widespread to remain expensive, the opportunity is simply too lucrative. Additionally, even if it does stay more expensive than some other drugs, it’s also worth considering that it’s the kind of drug people are probably willing to pay a bit more for.
The Reflexive Nature of a Successful Weight Loss Drug
The exponential rise in search trends for terms related to these drugs (GLP-1, Wegovy, Mounjaro, Ozempic) speaks to a reflexive relationship that is catalyzed by the nature of social media.. Since the re-release of Wegovy in 2021, both have rapidly become the talk of the digital town, turning the wheel of a virtuous cycle that has propelled them into the global spotlight.
Wikipedia Trends for “Semaglutide”:
Wikipedia Trends for Tirzepatide, which had a coupon program around its release in early Summer 2022. Despite the program ending, it rebounded in interest:
Google Search Trends for various GLP-1 Drugs:
The data paints an intriguing picture. According to Google Trends, searches for "Wegovy" and "Ozempic" have skyrocketed since 2021. In fact, the increase hasn't been linear—it's been exponential. From curiosity-seekers trying to understand these new options to individuals actively seeking weight-loss solutions, a wide swath of internet users have been part of this surge.
But why the sudden interest? The answer lies in the confluence of science and social media. People are no longer just passive consumers of health information. They're turning into active participants in a global conversation about weight loss and health
Picture this: A patient stumbles upon a TikTok video of someone sharing their positive experience with Wegovy. Inspired, she tries the drug and finds it works for her. Then, she posts her before-and-after photos on Instagram, attributing her success to Wegovy. Her followers—moved by her story and noticeable transformation—start their research, with "Wegovy" and "Ozempic" becoming their keywords. Consequently, the search trend climbs. And as more people share their experiences, this reflexive cycle continues, spiraling the search trend upward. Maybe she’s prescribed Ozempic instead, or Mounjaro. This sparks discussions on social media surrounding pharmaceutical solutions that don’t really happen (outside of illicit drugs), to the point where Ozempic was mentioned at the Oscars in 2023.
The significance of this trend extends beyond the digital realm. The popularity of Wegovy, Mounjaro and Ozempic on social media platforms is spurring real-world effects. It's shaping people's perceptions about weight loss, transforming attitudes towards obesity treatments, and most importantly, it's driving demand.
The Beneficiaries of GLP-1 Drugs Across Industries
Health Insurers and the Cost of Obesity
Source: McKinsey4
Although weight loss has been the main focus in the analyses of these drugs so far, it is important to bear in mind that with them comes the improvement in other measurements of health, such as blood glucose levels, cardiovascular parameters, cholesterol and triglyceride levels.
Source: Novo Investor Day 2022
Estimating the costs associated with diseases are inherently imprecise and noisy due to difficulties in normalising the difference between individual circumstances and lifestyles, but for the sake of providing ball-park figures, some studies have found that excess medical spending on obesity for adults is around 260 billion USD or around 1800 USD excess per person in 2019. Conservative estimates for the medical costs associated with overweightness and obesity in China have been projected to 61 billion USD. Obesity is obviously not restricted to the US; another study in has estimated that costs in Australia, Brazil, India, Mexico and Spain range from 20-40 billion dollars in 2019. .
Costs associated with obesity compound, since obese patients also become more susceptible to other diseases. The highest comorbidity (Presence of two or more health conditions at once in the same person) associated with obesity is T2DM. Diabetes is associated with retinopathy (Which can lead to problems in vision), nephropathy (Kidney problems) and coronary heart disease. Direct costs for treating diabetes globally have been estimated to be around 860 billion USD per year, and 150 billion USD per year in the U.S..
Obesity also increases the risk of cardiovascular disease, where 5 kg increases in weight in adults is associated with an 11% higher risk of death from cardiovascular disease (strokes, coronary heart disease, myocardial infarction etc…). An increased risk of NAFLD (Non-alcoholic fatty liver disease) is seen in obese patients, with annual burdens in the treatment of NAFLD estimated to be in the hundreds of billions for the US[10]. Specific forms of cancer are seen at a higher incidence and are associated with poorer survival rate in obese patients, including colorectal, breast, kidney, esophageal adenocarcinoma, endometrial and gallbladder cancer ,[12],[13]. Consequences of obesity also extend to psychological effects, where depression and anxiety have been shown to be associated with obesity[14]. With the caveat that not all people with diabetes, cardiovascular disease, NAFLD or cancer are obese, reductions in the prevalence of obesity would be expected to also reduce the prevalence of these diseases as well.
Outside of direct medical and health insurance costs, improvement of health alleviates drags on economic productivity by reducing death, days absent from work and disabilities (from joint issues). Among the overweight, a 10% reduction in body weight lowers lifetime medical costs by nearly 35%, while going from obese to overweight can result in halving the costs over a lifetime. A 2021 report published by the National Library of Medicine revealed that adult obesity is associated with average annual increased medical costs per person of $1,861. And severe obesity was associated with additional costs of $3,097 per adult.
Again, currently, the obesity rate in the U.S. is currently almost 43%. Using some naive assumptions here (that the average weight loss required for the average obese person’s BMI to be below 30 and thus not categorized as obese, assuming x% of obese people take the drug and all experience the average reduction in body weight), we can see a picture emerge that shows the potential to disrupt a trend that’s been forming for the past 50 years. At a 50% penetration rate, a 68-week course of Mounjaro would (in this ideal, naïve assumption) bring the obesity rate to the lowest level it’s been since 1981. Assuming everyone took it, made no lifestyle changes and gained back more than half of the weight after discontinuation with no use as a maintenance drug afterwards, utilizing a very pessimistic penetration rate of 15%, even then it would be enough to have the obesity rate correct lower by more than 6%.
That would be the first downtick in a generation…
While the benefits to mortality and, by extension, life insurers may be apparent it’s important to consider how expensive these drugs will be in the short term. We could see companies like Insperity (NSP) take a serious hit on the popularity of GLP-1 drugs.
I also think it’s important to give credit to just how skilled life insurance companies are at estimating when you will die. Life insurers can indeed adjust your premiums based on your height, weight and comorbidities. Health insurers, however, cannot. Under the Affordable Care Act (ACA), health insurance companies can't deny you coverage or charge you higher premiums on the basis of weight.
Once the data is sufficiently acceptable the cost-benefit analysis for the insurers is impossible to ignore they will begin covering these drugs widely, and the benefit to the patients will show up in terms of savings. Of course, there will be complex interplay and it won’t at all be pleasant (this is health insurance in the U.S., I don’t know what else you’d expect?) but overall it will be a net positive in the longer term. Whether the market looks past that in the short term…I’d say not.
Genuinely effective obesity drugs, as GLP-1s appear to be, should have a positive impact on the bottom line of US health insurers like Cigna (CI), United Healthcare (UNH), Humana (HUM) and Centene (CNC) in the long term while harming companies paying for these very expensive and likely to be in-demand medications like Insperity (NSP) and Robert Half (RHI).
The economic implications of obesity and diabetes are significant both in the real economy and in the revenue of large companies, with $1 out of every $4 in US health care costs is spent on caring for people with diabetes.
Furthermore, significant weight loss can tend to lead to complications that require cosmetic surgery such as abdominoplasty, breast reduction/augmentation and gluteal lifts to name a few. With the percentage of patients undergoing efforts at weight loss who are likely to be successful, there is a significant likelihood of increased business for a company like InMode (INMD) which sells products used to improve muscle tone after abdominoplasty or Establishment Labs (ESTA).
Weight Watchers (WW)
Source: SimilarWeb
WeightWatchers is a company with a concerning balance sheet that may potentially continue to catch a tailwind from GLP-1s due to one smart decision: acquiring Sequence Health. Sequence is a telehealth provider solely focused on GLP-1 prescriptions. What would have originally probably been the death knell for this long suffering company now may become its turnaround story.
With Sequence, WeightWatchers International is in possession of a database of email addresses and contact information not just for people who are overweight or want to lose weight, but of people who are overweight and have demonstrated the fact that they will pay money to change that. Now, that list has value considering the synergy in which they are converted to Sequence customers (and given something that is very likely to work).
WW is not something I’d bet the farm on, but it is reminiscent of other highly operationally levered companies in industries that experience sudden and sustained tailwinds like UAN, RIG or (more recently) AAOI. Honestly, WW probably still goes bankrupt but in the meantime the GLP-1 narrative can lift the stock to squeezy heights. It’s been struggling, for sure, but this might be the tailwind it needs to make it attractive again, especially with up to 6 million lapsed members that have expressed willingness to pay to lose weight and who could be brough back in by marketing campaigns like this below (combined with the general social media reflexivity noted above with GLP-1 drugs):
Dating Sites & Apps
It’s perhaps uncomfortable to confront but with nearly a third of every single person in the U.S. over 30 BMI (combined with the fact that the majority of them do not want to be) and being slim / in-shape viewed as generally attractive, it’s likely that successful weight loss drugs increase demand for dating apps like Tindr, Bumble, Hinge etc. Obesity has been found to lead to higher rates of depression and isolating behavior - something that is not conducive to dating. While I already like MTCH from a valuation standpoint, I think that this provides another tailwind for both Match Group (MTCH) and Bumble (BMBL) simply from people feeling better about the way they look and being more open to dating.
Clothing, Apparel and Fashion
When exploring the second-order effects of a reduction in the astronomical obesity rate, it’s natural to stop at the plus-size clothing retailers. Companies like Destination XL (DXLG) or Torrid (CURV). On twitter when I mentioned I was writing this article, some suggested they’d be shorts. I believe it’s a bit more complex than that. As we’ll see later when we go into the data, this still is not a magic shot - it’s worth noting that it still takes nearly a year for these outcomes to happen (and likely many patients will be n “maintenance doses” as they continue to lose weight beyond those timeframes). If you lose 20% of your body weight in a year, you’re going to be doing a lot of clothes shopping unless you want to look like someone threw a sheet on top of you and you decided that was dressing. I think that, especially in the beginning (right now) as these drugs are gaining notoriety and seeing increased usage, it’s tough to say but in the short-term, those plus-size clothing stores may actually see an increase in demand. As people who are losing weight need to purchase new clothes that fit their changing bodies, this could result in a temporary surge in revenue for clothing retailers, including plus-size specialists who often cater to a broad range of sizes. Additionally, as noted earlier, there is a risk that there is a rebound in weight.
Again it’s worth reiterating that the potential patient population for these drugs is 42% of the population. Trends in consumer purchases have been accomplished with much less.
The Victims of GLP-1 Drugs Across Industries
Figure 2, Average Weight Loss Across Treatments (%)
Diabesity-Related Surgical & Medical Devices
Sleep Apnea
Obstructive sleep apnea (OSA) is a common problem among obese patients (around 40% of obese patients suffer from OSA), while around 70% of OSA patients are obese[15]. 20% decreases in body weight have been associated with halving in the severity of sleep apnea metrics (AHI)[16]. Decreases in OSA would be expected to lead to improvements in sleep quality and thus mood and productivity.
Let’s look at the remission data for sleep apnea and some other chronic conditions after various bariatric interventions:
LSG (Sleeve Gastrectomy) resulted in a 75.8% remission rate for sleep apnea. Sleeve Gastrectomy is only marginally more effective, on average, than what we’ve seen above with Retatrutide. Obese patients comprise 41% of all Total Knee Arthroplasty surgeries in the US and roughly 3/4ths of all patients receiving CPAP therapy for sleep apnea. Additionally, CPAP compliance is typically not excellent. I believe that sales for a company like ResMed (RMD), within 5 years, could be cut in half - and there will be very little they can do about it. Lily’s SURMOUNT-OSA trial will begin in March 2024 to evaluate the effect of Mounjaro on sleep apnea. Most of the overall revenue from CPAP machines is not made on the machine itself, but on the provision of things like masks, tubes and humidifiers which need to be replaced regularly. There is simply no way that these drugs do not end up harming both the top and bottom line for these companies, as the expected duration of OSA decreases alongside the expected percent of the population suffering from it. Inspire Medical Systems (INSP), AdaptHealth (AHCO) & Quipt Home Medical (QIPT) all, additionally, face challenges.
For the medical device industry, the consequences of the GLP-1 revolution could be devastating indeed. Yet ResMed still trades at a 36x P/E.
Koninklijke Philips NV (PHG) has invested significantly in a rebuild of its Respironics division, which suffered a recall. You can see here, it expects sales to return to pre-recall levels as a result of those efforts by 2025. PHG is up 44% Year to Date as the writing of this piece.
Figure 3, Source: PHG Earnings Report
To be as endearingly direct as the Dutch tend to be - I think not. I personally believe that, of all the potentially negatively affected companies here, the CPAP machine ones are by far at the most risk.
This scenario analysis done by Morgan Stanley on the potential impact is staggering if you consider the arguments laid out earlier about the reflexive nature of a successful weight loss drug and the eventual deflation in popular pharmaceuticals to be accurate.
It cannot be underestimated how momentous something like this, if easily accessible, could be.
Why? Because it’s extremely unlikely that it occurs as is. During a maximum of 9 years’ follow-up, 1283 men and 2245 women attained normal body weight. In simple obesity (body mass index = 30.0–34.9 kg/m2), the annual probability of attaining normal weight was 1 in 210 for men and 1 in 124 for women, increasing to 1 in 1290 for men and 1 in 677 for women with morbid obesity (body mass index = 40.0–44.9 kg/m2). The cold, hard fact of the matter is this:
Without a solution like GLP-1 drugs, the obesity rate will likely continue climbing. Nobody wants to be obese, just like nobody wants to have erectile dysfunction, the hedonic desire to look good is something that should be factored in here. I think that once Mounjaro is approved for weight loss at the end of 2023 this trend will gain steam in a significant way, which would likely reflect in Q2-Q3 2024 earnings for PHG, RMD etc.
There are similar concerns for companies that make much of their money due to the obesity epidemic. This includes companies that provide diabetes related medical devices as well as orthopedic devices (total knee replacements are much more common in obese patients). This includes Tandem Diabetes (TNDM), Insulet (PODD), Dexcom (DXCM), Conmed (CNMD) et cetera.
It’s not just about diabetes treatment either, it’s about the impact of reducing the prognosis for those with obesity who have developed prediabetes and will, without intervention, develop Type 2 Diabetes. Mounjaro Ph3 trial SURMOUNT showed 95.3% of patients on Mounjaro with prediabetes at baseline reverted to normogylcemia by week 72.
Looking here we can see that Retatrutide results are only about 4-5% shy of Sleeve Gastrectomy, an honest-to-god surgical procedure requiring general anesthesia!!
This will likely make it very difficult going forward for companies relying on increased bariatric surgery volumes for growth, like Teleflex (TFX) or Allurion (ALUR).
Fast Food and Junk Food
Although difficult to measure precisely, since measuring individual consumption of fast food requires self-reporting, a questionnaire in Michigan showed that the prevalence of obesity rises from 24.1% in people who consume fast food less than once a week to 32.2% in people who consume fast food twice a week[18]. Other meta-analyses of cohort studies show that there tends to be an association between fast-food consumption and weight gain, although it is difficult to parse out the direction of causality from these studies alone[19].
On a less precise & scientifically accurate note, I consider the following - when we look into the revenue breakdown of companies that sell alcoholic beverages, a somewhat disturbing trend becomes apparent: it is alcoholics and binge drinkers who, despite making up a minority of the population, make up a significant portion of consumption and revenue:
I think it is not far-fetched to consider that the trends in the fast food industry are similar. Obese persons are likely responsible for an outsized percentage of sales by volume, and it’s not uncommon to meet someone with obesity who consumes fast food twice a day. Anecdotally, from my endocrinologist expert, Mounjaro tends to create an almost visceral aversion to fast food. I think it’s not unreasonable to assume that among fast food restaurants, widespread access and use of these drugs and future drugs for weight loss in this category will result in a threat to revenues. Mind you, I am not being optimistic here, this is not a “everyone will make better choices in their diet” assessment. This is, simply put, lower volumes. You just can’t spend $40 a day at a fast food joint if you’re eating 500 calories before you feel sick. I’m almost certain it would be literally impossible.
It’s easy to see an executive of a fast food company in the future, on an earnings call, with something along the lines of “GLP-1s affect sales? No, people eat at Heart Attack Burger because they enjoy our quality ingredients, friendly service and fresh preparations!” or something. I don’t think I need to go into why it’s not a stretch to imagine someone who is, quite literally, pathologically consuming this food to develop an aversion to it once the cycle is broken.
I believe I have collected an assortment of publicly listed fast food companies that would suffer most from this volume reduction and potential behavioral aversion in their most significant customers. They are YUM Brands (YUM), Domino’s Pizza (DPZ), Brinker International (EAT), Restaurant Brands (QSR), Potbelly Corp (PBPB), Red Robin Gourmet Burgers (RRGB) and Jack in the Box (JACK). While it may or may not belong here I think it also prudent to mention DoorDash (DASH) due to a business model that likely thrives on demand for fast food, although I don’t have any breakdown to support this, so I won’t be including it in the basket.
Health and Wellness
Planet Fitness’ (PLNT) business model and demographic is very specific. If you’re very passionate about going to the gym and physical fitness, you probably don’t work out at Planet Fitness. In fact, PLNT is more so in the business of selling subscriptions that only get used once or twice than it is in the business of fitness. From a pragmatic point of view, a successful weight loss drug that doesn’t require you to diet and exercise probably isn’t great for all the guilt sign ups powering PLNT’s ability to make a buck (although I fully support people changing their diet and exercise, there is the question of “if they were going to do that they wouldn’t have needed the drug in the first place”...). In a similar manner, products which serve the sole purpose of helping you to lose weight don’t really have a big place in this new paradigm, so a company like MediFast (MED) or Herbalife (HLF) could have issues.
The Science & The Future of GLP-1 Drugs
Peptides: GLP-1
Peptides are small molecules, essentially short proteins, that can be used for a variety of purposes in the body. Peptides from one part of the body can be transmitted to another part of the body to transmit information. Inside the gut, different types of peptides can be released to signal to the rest of the body that certain events are occurring (such as detecting when food is coming into the intestine). Relaying of this signal to other parts of the body, such as the liver, pancreas or brain is then required in turn to engage appropriate responses, such as getting the body ready to digest the food, prepare insulin release and to make us feel full.
GLP-1 (Glucagon-like peptide-1) is one such peptide, released by the intestine, which helps prime insulin release, thus allowing the body to absorb glucose after a meal. Specific pancreatic cells are able to recognise GLP-1 through the use of molecules called receptors (Specifically the GLP-1 Receptor: GLP1R), which in turn help ‘decipher’ the message. When specific cells are capable of recognising GLP-1 through the use of GLP1R, the technical term is to say that they “express GLP1R” The net result is that the pancreas begins to secrete insulin after receiving GLP-1, thus ensuring that insulin release occurs shortly after a meal, which is required for normal healthy functioning.
Peptides eventually disappear over time through natural processes, meaning that they have a limited life-span; to ensure that the body does not mistakenly keep signals on for too long (An easy example to consider is when you feel full after a meal; this feeling of fullness disappears over time so that you can get hungry again). The time it takes for a peptide can be measured by its “half-life”, which mathematically is defined as the time it takes for half of the initial amount of peptide to disappear.
Among other benefits, GLP-1 is able to reduce blood glucose levels via insulin release, a highly desirable effect in patients with diabetes. However, GLP-1 is only able to create these effects for a short time-span due to the fact that it disappears over time. By engineering drugs that mimic these hormones (called “agonists”) with a longer half-life; the desirable effects of GLP-1 can be sustained over time. Such logic formed the basis for the initial development of GLP1R agonists.
Central action of GLP-1
GLP1R agonists were predicted to prolong the feeling of fullness/satiety, via the slowing of gastric emptying, which is an effect of GLP1R in the gut. However, GLP1R agonists produce a variety of effects in the brain as well, making efforts to increase the ability of such drugs to cross the blood-brain barrier of high interest. Literature on this topic is too large to summarise comprehensively, but it suffices to say that GLP1R agonists are capable of increasing the activity of neurons that promote satiety, while simultaneously decreasing the activity of neurons that promote hunger.
The effect that the brain can have on appetite has been long known; a classical experiment involving damaging a small part of the brain (In technical jargon, a bilateral Ventromedial hypothalamic lesion) caused rats to become morbidly obese (Pictured to the right). Moreover, the common side-effect of GLP1R agonists in producing nausea is attributed to GLP1R-expressing neurons in the Area Postrema[20], which is well known to regulate processes such as vomiting in humans.
The effects of GLP1R agonists is not restricted to the brain, as it also has direct effects on the neurons that receive information from the gut (Vagal neurons)[21]. In these neurons, the presence of GLP-1 signifies mechanical distension in the gut, signalling the neurons to facilitate increases in gastric pressure which is needed for normal digestion and may explain other gastrointestinal side-effects such as diarrhea.
Currently Approved GLP-1 Drugs
Novo Nordisk: Semaglutide (Ozempic/Wegovy/Rybelsus)
Semaglutide differs chemically from GLP-1 only slightly, yet this small difference allows semaglutide to have a half-life of around 7 days in the blood, making it a longer-lasting GLP-1. Semaglutide was originally intended for use in diabetic patients through subcutaneous injections. However, it quickly began apparent that one notable effect was the ability of semaglutide to also cause decreases in weight.
In a double blinded randomised study, daily injections of semaglutide (0.4 mg) in obese patients resulted in an average of 14% decrease in body weight, over placebo, after a year. In contrast, another GLP1R agonist, with a shorter half-life of around 13 hours (Liraglutide) achieved only 5.5% reduction in the same time span[22], demonstrating that improving half-life can result in better outcomes.
In a separate 2 year double blinded, placebo-controlled study, weekly subcutaneous injections of semaglutide (2.4mg) in obese patients, average body weights decreased 12.6% over placebo at the 2-year point[23]. 77.1% of patients on semaglutide achieved more than 5% weight loss, compared to 34.4% on placebo.
These decreases in body weight were not without costs, as semaglutide frequently caused gastrointestinal related side effects in patients. Nausea (53.3% of patients), diarrhea (34.9%) and vomiting (30.3%) being a few of the most common. Semaglutide injections are approved for weight loss by the FDA.
Dual GLP1R/GIPR agonists
GIP (Glucose-dependent insulinotropic polypeptide) is another peptide which is capable of increasing insulin release. This effect is mediated by GIP receptors (GIPR) expressed by pancreatic cells.
By creating drugs that can act as both GLP1R agonists and GIPR agonist, (Dual agonist for short), the original aim was to create either additive or synergistic effects on insulin release for treating diabetes. Interestingly, GIPR agonists alone do not typically result in weight loss. However, among other effects, it is in part able to suppress nausea, by acting directly on the Area Postrema, (the same part of the brain that we introduced which regulates vomiting in humans)[24], which is thought to be a contributing factor as to why dual GLP1R/GIPR agonists are able to act synergistically. Due to this effect, patients are able to take GLP1R agonists, when paired with these compounds, at higher doses without the side effects becoming too severe (increasing therapeutic efficacy).
Eli Lilly: Tirzepatide/Mounjaro
Lily initially developed Tirzepatide as a dual agonist, with a half-life of around 5 days, for the treatment of T2DM. Although a dual agonist capable of activating both GLP1R and GIPR, Tirzepatide preferentially activates GIPR more, over GLP1R. In a 40-week double blinded, placebo-controlled study, weekly injections of Tirzepatide (15mg) in Type 2 Diabetes Mellitus (T2DM) patients was capable of reducing body weight by an average of 10.1%, over placebo at 40 weeks.
77% of patients achieved more than 5% weight loss compared to 14% on placebo.
47% of patients achieved more than 10% weight loss compared to 1% on placebo.
27% of patients achieved more than 15% weight loss compared to 0% on placebo.
Side effects on Tirzepatide are qualitatively similar, but less frequent compared to Semaglutide. Side effects are nausea (18% of patients), diarrhea (12%) and vomiting (6%)[25].
In a separate double blinded placebo-controlled trial, weekly injections of Tirzepatide (15mg) in conjunction with lifestyle changes in obese patients could decrease body weight by 17.8% on average, over placebo after 72 weeks.
96.3% of patients achieved more than 5% weight loss compared to 27.9% on lifestyle changes alone.
90.1% of patients achieved more than 10% weight loss compared to 13.5% on lifestyle changes alone
78.2% of patients achieved more than 15% weight loss compared to 6% on lifestyle changes alone.
62.9% of patients achieved more than 20% weight loss compared to 1.3% on lifestyle changes alone. 39.7% of patients achieved more than 25% weight loss compared to 0.3% on lifestyle changes alone.
Side effects included nausea (31% of patients), diarrhea (23%) and vomiting (12.2%)[26].
Taken together Tirzepatide shows that a preferential activation of GIPR over GLP1R is capable of producing large decreases in weight without severely increasing side effects. This appears largely due to the ability of GIPR agonism to suppress the nausea caused by GLP1R agonism. Mounjaro is more effective and with less side effects than Ozempic/Wegovy, but also can result in increased muscle mass loss due to this efficacy.
Mounjaro will likely be approved for the weight loss indication in Q4 2023.
Novo Nordisk: Rybelsus
Rybelsus sidesteps the need for injections, being an oral form of Semaglutide. In a double blinded placebo controlled phase 3 study, obese patients were treated with either daily Rybelsus (50 mg) or placebo for 68 weeks. Mean body weights decreases of 12.7% over placebo were seen after 68 weeks in Rybelsus treated patients.
85% of patients achieved more than 5% weight loss compared to 26% on placebo.
69% of patients achieved more than 10% weight loss compared to 12% on placebo.
54% of patients achieved more than 15% weight loss compared to 6% on placebo.
34% of patients achieved more than 15% weight loss compared to 3% on placebo.
Side effects were qualitatively similar with semaglutide, as expected: nausea (52% of patients), diarrhea (27%) and vomiting (24%) being a few of the most common. [27]
Interestingly, the oral form, Rybelsus, appears to be slightly better than semaglutide in terms of improving body weight, when considering the shorter time it requires to achieve a 12.7% decrease in body weight (68 weeks) vs semaglutide injections (2 years). Rybelsus is FDA approved, but not yet approved for weight loss.
Eli Llly: Orfoglipron/LY3502970/ Formerly Chugai OWL833
Orfoglipron is an oral GLP1R agonist, taken daily due to its half life of 25-68 hours[28]. A double blinded placebo controlled study using daily Orfoglipron (45 mg) in obese patients, found an average of 12.4% decrease in body weight over placebo after 36 weeks.
90% of patients achieved more than 5% weight loss compared to 24% on placebo.
69% of patients achieved more than 10% weight loss compared to 9% on placebo.
48% of patients achieved more than 15% weight loss compared to 1% on placebo.
Side effects included nausea (39% of patients), diarrhea (25%) and vomiting (28%)[29]
Overall, orfoglipron achieves comparable weight loss with other GLP1R agonists such as semaglutide and Rybelsus, but has a slightly better side effect profile and achieves weight loss in a shorter period of time. It is not currently FDA approved.
Clinical-Stage GLP-1 Drugs
Fat mouse, lol.
Eli Lilly: Mazdutide/ LY3305577 / Formerly Innovent IBI362
Oxyntomodulin (OXM) is a peptide which acts as a dual GLP1R/GR dual agonist. Mazdutide, was designed by Innovent (Originally IBI362) to mimic OXM but had chemical modifications to extend it’s half-life, to around 7.5-9.8 days[30]. A double-blinded placebo-controlled phase 2 clinical study in obese patients weekly injections of mazdutide (9 mg) could cause 15.4% decreases in body weight over placebo after 24 weeks.
81.7% of patients achieved more than 5% weight loss compared to 0% placebo.
65% of patients achieved more than 10% weight loss compared to 0% placebo.
31.7% of patients achieved more than 15% weight loss compared to 0% placebo.
21.7% of patients achieved more than 20% weight loss compared to 0% placebo. [31]
Proportions of side effects were not noted in the phase 2 study, but results from an earlier phase 1 study, with 8 obese patients receiving weekly Mazdutide (9 mg) showed side effects of nausea (62.5% of patients), diarrhea (25%) and vomiting (37.5%) were seen after 12 weeks [32], although it must be noted that these numbers probably deviate largely from the results in the phase 2 study, simply due to sheer differences in sample sizes.
Mazdutide overall appears to achieve superior weight loss, in shorter periods of times compared to GLP1R agonists. However, due to the lower sample sizes, these impressive effects are likely to become more tempered as clinical trials progress.
Glucagon Receptors and Triple Agonists (GGG)
While GLP1R and GIPR reduce caloric intake, by reducing appetite, Glucagon receptors (GR) can act in concert by increasing energy expenditure. Brown adipose tissue (BAT; A type of fat cell) express GR and respond to glucagon by increasing energy expenditure occurs via thermogenesis (Essentially, burning fat to produce heat). This has the additional advantage of reducing body weight by reducing percentage fat in the body. However, GR also is involved in increasing blood glucose (Undesirable in diabetic patients), meaning this needs to be offset by corresponding decreases in blood glucose via GLP1R. Achieving the right balance between GLP1R, GIPR and GR agonisation, would in principle be able to achieve even greater losses in weight, while still preserving lowering of blood glucose.
This balancing act was attempted by Novo Nordisk, when they developed a triple agonist, compound 16, which in mice was able to reduce body weight by an average of around 30%, fat mass by around 55% and lean mass by around 10% after 27 days[33]. Novo Nordisk have however since discontinued their research in triple agonists, citing their intention to focus on semaglutide.
(One should bear in mind that results in mice are going to be more dramatic compared to humans, due in part to higher dosages. These higher dosages are possible to achieve in mice, since they cannot complain to scientists about side effects occurring at high dosages and do not have the physical capability to prevent themselves from receiving drug injections)
Sanofi produced a triple agonist, SAR441255, which was capable of decreasing body weight in mice by around 14% at 26 days, 12% in monkeys by 42 days[34], although Sanofi have since discontinued development of the triple agonist for human use for uncited reasons.
Hanmi Pharmaceuticals: Efocipegtrutide/HM15211
Currently undergoing phase 2 trials for NASH patients, Hanmi Pharmaceuticals have also produced their own triple agonist, efocipegtrutide/HM15211. In mice, efocipegtruride has been shown to be able to reduce body weight by 43% after 4 weeks of treatment. Worth mentioning is that the degree of reduced food intake between efocipegtrutide and liraglutide (A GLP1R agonist) treated mice did not differ[35], demonstrating that the enhanced weight decrease was occurring through enhanced energy expenditure. During phase 1 trials in NASH patients, efocipegtrutide showed a half-life of around 5.9 days, and was capable of reducing body weight by 5.1% when administered weekly at 0.08mg/kg, after 12 weeks[36].
Eli Lilly: Retatrutide/ LY3437943
Eli Lilly currently have clinical trials running for their triple agonist, Retatrutide/LY3437943. Retatrutide, like Tirzepatide, maintains a heavy preference for activating GIPR over GLP1R, while its ability to activate GR is much lower compared to both. A half-life of 5 days also allows Retatrutide to be administered only weekly[37]. In a double blinded placebo-controlled study, weekly injections of retatrutide (12 mg) in obese patients, in conjunction with lifestyle changes, showed average weight decreases of 22.1% over placebo patients after 48 weeks. Weight loss was more pronounced in patients with a BMI over 35, compared to those under 35.
100% of patients achieved more than 5% weight loss compared to 27% on lifestyle changes alone.
93 % of patients achieved more than 10% weight loss compared to 9% on lifestyle changes alone
83% of patients achieved more than 15% weight loss compared to 2% on lifestyle changes alone.
63% of patients achieved more than 20% weight loss compared to 1% on lifestyle changes alone.
48% of patients achieved more than 25% weight loss compared to 0% on lifestyle changes alone.
26% of patients achieved more than 30% weight loss compared to 0% on lifestyle changes alone.
Side effects included nausea (16% of patients), diarrhea (2%) and vomiting (2%)[38].
In the Retatrutide trial, it was noted: “this is an unusually high magnitude of efficacy as compared with findings in clinical trials of other anti- obesity agents, although it has been observed with bariatric–metabolic surgery.”
Basically, if we could give everyone in America bariatric surgery, we could probably solve the obesity epidemic. But not everyone wants surgery, and that’s extremely expensive. Considering the possibility of bariatric surgery in a pill/autoinjector is quite a thing.
These results taken together suggest that adding even mild amounts of GR agonism (to increase energy expenditure) on top of a dual agonist like Tirzepatide is able to produce even more pronounced decreases in weight.
Different combinations of dual agonist: GIP1R and GR
Altimmune: Pemvidutide/ALT-801
Pemvidutide is a dual agonist, but for GLP1R and GR, instead of GLP1R and GIPR. Pemvidutide is a balanced dual agonist, meaning that it does not preferentially activate GLP1R or GR over the other[39]. In a double blinded, placebo-controlled trial, patients showed an average weight loss of 9.9% at 24 weeks on weekly pemvidutide (2.4mg) over placebo.
84.6% of patients achieved more than 5% weight loss compared to 25% on lifestyle changes alone.
50% of patients achieved more than 10% weight loss compared to 0% on lifestyle changes alone
23.1% of patients achieved more than 15% weight loss compared to 0% on lifestyle changes alone.
Side effects included nausea (53.7% of patients), diarrhea (14.7%) and vomiting (24.4%). Worryingly however, 26.8% of patients had to discontinue the trial due to the severity of side-effects, although it must be noted that this was in part due to the lack of a run-in (A time frame where drug dosage is started from a low dosage and then escalated over time until the highest tolerable dose for the patient is reached)[40].
Taken in conjunction with previous results, it suggests that while GLP1R and GR agonism in conjunction is able to produce large decreases in weight (Via the reduction of food intake and increased energy expenditure), but the absence of GIPR agonism produces a more unfavourable frequency of side-effects. Trial results at the 48 week mark are expected by 4Q 2023.
Novartis: Bimagrumab/BYM338
Bimagrumab is an antibody against activin type II receptor (ActRII) meaning that it acts as an ActRII antagonist (It prevents ActRII from functioning).
In a double blinded placebo controlled phase 2 study, patients with T2DM and obesity were given intravenous bimagrumab (once every 4 weeks, to a maximum dosage of 1200mg ) in combination with diet and exercise counselling. Total fat mass was shown to have decreased by 20% over placebo, whereas lean mass increased by 4.4% over placebo, in bimagrumab treated patients after 48 weeks.
96% of patients achieved more than 5% fat mass loss compared to 21% on diet and exercise counselling alone.
92% of patients achieved more than 10% weight loss compared to 10% on diet and exercise counselling alone.
77% of patients achieved more than 15% weight loss compared to 10% on diet and exercise counselling alone.[41]
Bimagrumab shows a separate mechanism from GLP1R or GIPR agonism, instead targeting muscle cells. Likewise, side effects from this drug are more specific to the muscles, including spasms, but its ability to increase lean mass at the expense of fat suggests that it may find use in combination with other drugs to ensure body weight loss is preferentially targeted towards fat, while ensuring muscle is spared.
Indeed, Versanis Bio have tried a combination of bimagrumab and semaglutide in a prior phase 2a study assessing bimagrumab in patients with obesity and type 2 diabetes. Patients showed a 21.9% reduction in fat mass after 48 weeks of therapy while increasing lean muscle mass by 4.5%. In the same study, patients were followed for an additional 12 weeks after the last dose of bimagrumab and experienced no weight regain. In contrast, rapid weight regain has been observed in patients who stop taking semaglutide and other incretins.
Novo Nordisk: CagriSema
Cagrilinitide is an analogue (a drug made to mimic a molecule already present in the body) of amylin, which acts as an agonist for calcitonin receptors. Amylin is also a gut peptide, which has a vast literature of showing weight decreases in mice[42]. In a double blinded phase 2 study, T2DM and obese patients were treated with CagriSema, Semaglutide alone or Cagrlinitide alone (All escalated to 2.4 mg). Mean body weight changes across the groups at the end of 32 weeks were:
CagriSema: 15.6%
Semaglutide alone: 5.1%
Cagrilinitide alone: 8.1%[43]
Side effects included nausea (29% of patients), diarrhea (16%) and vomiting (10%) in CagriSema treated patients. These results suggest that GLP1R agonists in combination with a calcitonin receptor agonist, is able to show even larger decreases in body weight than typical GLP1R agonists, within a shorter time and with a more favourable side effect profile.
Boehringer Ingelheim / Zealand Pharma (ZLDPF): Survodutide (aka BI 456906)[44]
In a placebo-controlled study evaluating the GLP1R/GR dual agonist (Obesity Indication), patients received weekly subcutaneous injections of 4.8mg for a duration of 46 weeks. The results showed a significant decrease in mean body weight, with a reduction of 12.1% compared to the placebo group. Remarkably, up to 40% of individuals who received the highest two doses of survodutide (BI 456906) achieved a weight loss of at least 20%. A phase 3 go/no-go decision is expected in 2023.
Figures and Graphs:
Average body weight decreases, percentage of patients achieving different body weight decrease outcomes, percentage of patients suffering from side effects and mean body weight loss, normalised for time.
Body weight loss percentage according to trial
Estimated body weight loss percentage, normalised to 24 weeks
More Speculative Peptide Drugs
It suffices to say that anti-obesity drugs have undergone large gains in outcomes, larger average percentage decreases in weight and reductions in side-effects.
Improvement and competition is expected over time, whether by creating more modifications to improve half-life, improving access to the brain, converting existing injectable drugs into oral forms, combining with other agonists or antagonists (Molecules that prevent receptors/molecules from functioning) such as DPP-4 inhibitors, which help extend the half-life of peptides such as GLP-1 and GIP.
Novo Nordisk are leaning heavily into their oral formulation of Semaglutide, Rybelsus, (Oral formulations face their own unique challenges due to the requirement to avoid extensive degradation of the drug by the digestive system), which although shows less weight loss compared to other competitors, has the convenience of not requiring injections. Aside from the advantage of having higher appeal to people who are squeamish with needles, oral formulations remove the need for cold chain storage for injectable forms of GLP1R/GIPR/GR agonists, which otherwise must be stored at cold temperatures if not provided in a dry (Tablet/pill) form. Whether Novo Nordisk remains the sole provider of oral weight-loss drugs, or if other companies begin to develop oral formulations of their own drugs is of high interest.
So far, Eli Lilly and Hanmi Pharmaceuticals are the only companies so far pursuing clinical trials of triple agonists. Whether Novo Nordisk or Sanofi decide to renew their efforts for a triple agonist, or other competitors decide to compete also remains to be seen.
So far preferential activation of GIPR activation, combined with less activation of GLP1R and even less activation of GR appear to have produced the most effective combination, as shown by Retatrutide, although whether this mixture is indeed the optimal is still an open question.
Although many other GLP1R agonists and GLP1R/GR dual agonists are in development, preliminary findings suggest that activation of GIPR is required to produce favourable side-effect profiles. Its combination with GLP1R agonists in particular is capable of producing large decreases in weight. Nausea, diarrhea and vomiting are the major side-effects that may deter patients from staying on medication, and taking higher doses of medication; consequently, the inhibition of these side-effects by GIPR agonism is critical.
DPP-4 (not described in detail in this article) is the enzyme responsible for degrading native GLP-1 and GIP in the body. By inhibiting DPP-4, one can enhance the half-life of GLP-1 and GIP already existing in the body. By itself however, decreasing the activity of DPP-4 has not shown any weight reducing effects[45], but whether its use in combination with other dual agonists/triple agonists remains to be seen. This route however appears to be largely ignored and probably will continue to be ignored, since the strategy so far has been to simply create agonists which are structurally designed to be resistant to DPP-4 (In fact, most modifications to extend the half-life of GLP1R/GIPR/GR agonists so far, do so by making the drugs resistant to DPP-4).
A plethora of other gut released peptides exist to be explored: Amylin[46], peptide YY (PYY)[47], Cholecystokinin (CCK), secretin, neurotensin, somatostatin (SST) and ghrelin to name a few. Some of these already have agonists. As mentioned in the case of CagriSema, natural amylin is a pancreatic hormone that induces satiety and Cagrilintide is a long-acting amylin analogue.
What is particularly notable about the CagriSema combination is the much better side effect profile while still retaining body weight loss, comparable to other GLP1R/GIPR dual agonists. Different combinations or even larger cocktails of agonists and/or antagonists may turn out to be even more effective going forward.
If met with success, this may provide impetus for further investment and research into utilising other types of peptides, not only to compete with current weight loss drugs available, but for other conditions and diseases that render themselves treatable through peptide agonists; chronic pain, migraines and bone disorders to name just a few. The information communicated by peptides is not limited to metabolic diseases, but also includes processes such as pain, immune modulation and cognitive effects.
Rhythm Pharmaceuticals (RYTM) is also involved in peptidergic drugs for weight loss. Its drug Imcivree (setmelanotide) has been approved by the FDA for three rare genetic diseases that cause obesity and is currently seeking approval for Hypothalamic Obesity (obesity due to genetic or physical damage to the hypothalamus). It has a neuroendocrine pathway independent of the GLP-1 receptors, working by stimulating melanocortin-4 receptors (MC4R) to reduce hunger and increase basal metabolic rate. The side effect profile is more serious than GLP1R agonists, including depression and suicidal ideation and it is not approved for other types of obesity. MC4R mutations are a rare form of genetic disorder that leads to obesity (Pictured to the right), reduced metabolic rate and glucose imbalances. This disease is readily treated by MC4R agonists, such as setmelanotide.
Potential downsides
Although current dual and triple agonists have not seen any long-term harmful effects so far, it is a possibility worth considering, given the history of anti-obesity drugs. Fenfluramine, one of the earliest weight loss drugs, was found over time to be associated with valvular heart disease[48] and was withdrawn from the market by the FDA. In more recent times, lorcaserin (Belviq) was also withdrawn, due to an increased risk (albeit only 9%) of cancer[49], which the FDA deemed as not being outweighed by the benefits of lorcaserin for any identifiable patient population. In 2008, Rimonabant (also known as SR141716) was withdrawn from the market worldwide after being approved in 2006 due to serious psychiatric side effects that outweighed its benefits, including suicide.[50]
However, it is important to note that fenfluramine, rimonabant and lorcaserin used very different molecular mechanisms (acting mainly on serotonin 2c receptors[51] in the hypothalamus [52] or CB1 receptors[53]) from GLP1R, GIPR or GR agonists and likewise do not imply anything on the possible dangers of GLP1R, GIPR or GR agonists.
Existing GLP1R agonists come with boxed warnings of thyroid cancer and pancreatitis, based mainly on rodent studies, where the effects of GLP1R agonism have noted species differences (For example, weight loss in rodents is mainly due to increased energy expenditure, whereas in humans it occurs via less food intake[54]). Although some studies have shown an increased risk for thyroid cancer and pancreatitis in human patients, these studies are difficult to interpret due to reporting bias, lack of controlling for other patient medical histories and comorbidities, the fact that obese people are already highly at risk for specific forms of cancer and may already have primordial tumours present when beginning medication and are likely to be taking other medication apart from GLP1R agonists as well [55]. Taken together these suggest that although GLP1R agonists may increase the risk of pancreatitis and thyroid cancer in rodents, that it has yet to be confirmed in humans.
The most likely cause of a withdrawal (If one were to occur) would probably be increased heart-rate caused by the agonisation of GLP1R and GR in particular [56],[57], effects that occur in part due to the expression of GLP1R and GR by the heart.
Although an increase in heart-rate per se is not a major problem for most people, it is salient in the case of GLP1R, GIPR or GR agonists since the target patient population of these agonist are obese patients, who likely already suffer from cardiovascular complications. However, so far across studies with GLP1R agonists, cardiovascular measurements such as blood pressure have seen improvements in general, with no significant increases in adverse cardiovascular effects seen in patients even after 2 years (In fact, decreases were seen), suggesting that the cardiovascular benefits of decreased body weight may overcome any potential harm that increases in heart rate by GLP1R agonists may cause. A phase 2 trial with Retatrutide also noted slight increase in heart rate (around 10bpm) after 24 weeks, but that this increased heart rate subsides over time. Thus, although an important point to keep attention to, data so far does not indicate that cardiovascular complications are a major cause for concern at the moment (gastrointestinal problems being by far the larger problem experienced by people on GLP1R agonists).
Development in the field is competitive and many companies are at varying stages, both clinical and preclinical, for new agonists to target weight loss. The sources for the data on these drugs are rather patchy, making direct comparisons difficult due to incomplete data. Baseline patient characteristics, full frequencies of side effects and study structure are not described in detail. We briefly touch upon some of the available information for upcoming drugs below, both in the US and abroad:
Tern Pharmaceutical (TERN): TERN-601
TERN-601 is a preclinical GLP1R agonist, which in mice has been shown to be able to reduce food intake, slow gastric emptying and improve glucose tolerance. Clinical trials are expected to start in the second half of 2023, with topline data due in 2024. TERN is also initiating IND-enabling activities in 2023 for TERN-800, an oral GIPR modulator, with the intention of being used in combination with GLP1R agonists.
Shandong Suncadia Medicine (Subsidiary of Jiangsu Hengrui Pharmaceutical) HRS-7535[58]
In a Phase 1 clinical trial conducted by Shandong Suncadia Medicine, a subsidiary of Jiangsu Hengrui Pharmaceutical, the GLP1R agonist HRS-7535 was investigated. The study employed a double-blinded design, with participants receiving a daily dose of 120mg HRS-7535 for a period of 12 weeks. Results demonstrated a remarkable 4.4kg decrease observed within just four weeks of treatment.
Fujian Shengdi Pharmaceutical: HRS9531 (Subsidiary of Jiangsu Hengrui Pharmaceutical)[59]
In a phase 1 study evaluating an oral GLP1R/GIP dual agonist, participants received subcutaneous injections of 8.1mg. Notably, the maximal mean weight loss observed was 4.9% after just 8 days of treatment. Furthermore, in the 5.4mg group, the maximum weight loss of 10% was achieved after 36 days.
Structure Therapeutics: GSBR-1290 [60][61]
An oral GLP1R agonist, currently being studied in a double-blinded, placebo-controlled Phase 2a clinical trial. Participants are receiving a daily dose of 120mg, and the study is expected to finish at the end of 2023. Preliminary data suggests an excellent pharmacokinetic (PK) profile. Additionally, a Phase 2b ethnobridging study evaluating the GLP1R agonist in Japanese individuals, named GSBR-1290, is planned to commence in 2024. These developments mark important milestones in the ongoing research and development of the oral GLP1R agonist.[62]
Viking Therapeutics (VK2735)
VK2735 is a GLP1R/GIPR dual agonist, with a half-life of around 170-250 hours, undergoing both oral and subcutaneous injection clinical trials.
In obese mice, VK2735 shows effects comparable to semaglutide, reducing body weight by up to 26.67% in 20 days. A Phase 1 data showed that weekly injections of VK2735 led to mean body weight decrease ranging up to 6% over placebo.
Zealand Pharma:
Dapiglutide/ ZP 7570[63]
Dapiglutide is a dual GLP1R/GLP2R agonist. Weekly injections of 6 mg resulted in a significant body weight loss of 4.5% after just four weeks of treatment. The dual agonist exhibits a half-life of approximately 123-129 hours, indicating a sustained duration of action.
ZP 6590[64]
ZP-6590 is a GIPR agonist, and has been designed to both reduce nausea and food intake. In mice, ZP-6590 was shown to be able to accentuate the body weight loss caused by semaglutide. ZP-6590 in combination with semaglutide produced mean weight losses of around 25% after 34 days in mice. Zealand Pharma: ZP 8396 (Amylin analog)[65]
In a randomized, double-blinded, placebo-controlled study, an amylin analog, ZP-8396, was investigated in a cohort of 56 male subjects, both lean and obese individuals. The drug exhibited a half-life of approximately 10 days. After one week of treatment, the mean body weight in the 2.4mg treated group decreased by 4.2% compared to the placebo group. Although some side effects, predominantly related to nausea and vomiting, were observed, they were generally mild and temporary in nature.
The GLP-1 Basket
If I were to weight this in a discretionary manner I’d overweight LLY and NVO by a significant amount and underweight the more speculative things that are higher risk/reward. If I were to play this theme as a Long/Short, which I think is possible because of the correlations between names that are affected positively and not positively.
This theme has progressed and so has our basket, meaning we have cut some losers as well as added to some winners while also adjusting which areas of the theme we are allocated to. Here is what it currently looks like in case anyone is reading after publication.
If you’d like to find out why we took profit on our MedTech shorts, or find out when we booked a +300% gain on CURV, read about how recent developments have informed our allocation etc. please consider upgrading to a paid subscription.5
Clinical Stage Basket (Equal Weighted)
Clinical bio is always gambling as far as I’m concerned, but it’s there if you want it. I’ll wait for some more inclinations about which way we are headed (and for a more forgiving environment for biopharma aka lower rates) to put any of these on.
Over the past year, the short and long basket actually have a decent correlation (0.779) so this could prove to be a good collection for L/S based on this single factor.
Paid subscribers have also received our view on being long smaller, clinical stage names like Gubra (GUBRA DC), Zealand Pharma (ZEAL DC) and Amgen (AMGN US). All of this is available to paid subscribers in our archive.
Lily and Novo already trade at rich valuations, but during the ramp of Viagra while there were still no generic alternatives Pfizer traded higher than 90x LTM earnings. Furthermore, I think there will be a phenomenon here where people are simply unwilling to accept the possibility of a weight loss drug without significant and disastrous side effects and that could contribute to good opportunities to buy panicky sell-offs in the primary names right now. For example, there is this:
There are definitely things to pay attention to as far as side effects go. Cardiovascular side effects in particular. These, however, are not them. Why? Because these are just people reporting the side effects associated with losing weight. As you age, you lose fat in your face. It makes your face look less full & more gaunt. The same happens when you are losing a lot of fat in general. When you are overweight or obese, your face naturally looks fuller. It is easier to notice wrinkles on a deflated balloon, so to speak.
In the same vein, there’s something called telogen effluvium which is stress associated hair loss that is temporary and very much reversible. Losing weight is not something your body wants you to do, it’s more or less an evolutionary fact that it is very much in support of you retaining excess calories to use as energy later when you are getting chased by a tiger or something. There are promising avenues of discovery in terms of retaining muscle, slowing down the weight loss so that the hair loss isn’t as severe et cetera. These are all side effects that would happen if you were to restrict yourself calorically to lose weight in a natural way.
There’s also this recent headline:
Which is a touchy subject and I don’t want to settle the European regulators investigation (notoriously not overly sensitive about things), but I’d venture to say that the psychological impact of obesity and the psychiatric side effects associated with changing body type including dysmorphia and eating disorders probably indicate that this is not a side effect of the drug but rather a side effect of the primary effect of the drug, in a small subset of patients.
So far, liraglutide and semaglutide alone have 20 million patient years of exposure. I think the reason we are seeing these increased reports of side effects (150 cases of self harm or suicidal ideation in the EU, which is a small number) is simply because more people are taking them and because people are mistaking the secondary effects of weight loss with the side effects of the drug. It’s worth monitoring these side effects reports, both because there still may be something genuinely concerning and because they may potentially present a good opportunity.
Obese people in general are more prone to suicidal ideation, making it difficult to attribute any suicidal ideation to drugs unless a study using a direct comparison between obese patients on and off GLP1R agonists is completed.[66][67]
Conclusion
GLP-1 Drugs work for their intended purpose and will likely represent a revolution in the way obesity is treated as well as obesity rates in the developed world. The easiest way to get exposure to this trend is with LLY and NVO, as their duopoly will maintain until at least 2027.
The consequences and benefits for various parts of the economy now that we are seeing outcomes previously only consistent with bariatric surgery available in a pharmaceutical will be wide-reaching and important for investors to monitor. These second order effects will become more far reaching as insurance coverage becomes wider for these drugs - this will be the differentiator between the risk/reward being better on the first-order beneficiaries (LLY and NVO) and the second-order plays like short bariatric surgery/sleep apnea/gyms/fast food & CPG.
The behavioral impact for the consumer if “anyone can be as slim as they want” will be equally important and potentially accretive to the total addressable market of more discerning luxury clothing and dating app companies while being relatively destructive to the bottom line of companies that rely on the American Overeater.
If you are uncomfortable with the current valuations of LLY and NVO (don’t be, they’re fine), I would recommend looking into more downstream effects where tailwinds provided by these drugs may not be priced in to expectations by the market and/or consensus estimates yet. I have provided some broad categories and a few names, but I’m sure there are more (if you find a good one, please let me know).
EDIT: Section added after original publication for release to free subscribers.
This could be a big deal for companies that are selling protein shakes but not marketing directly for “weight loss”, products like Fairlife or Premier Protein ready-to-drink shakes could see big volume increases potentially as market penetration increases and insurance coverage becomes more broad.
MEDICAL REFERENCES [1https://www.sportanddev.org/sites/default/files/downloads/mgi_overcoming_obesity_full_report_2_.pdf
[2] https://investor.novonordisk.com/q1-2022-presentation/?page=139
[3] Cawley, J., Biener, A., Meyerhoefer, C., Ding, Y., Zvenyach, T., Smolarz, B. G., & Ramasamy, A. (2021). Direct medical costs of obesity in the United States and the most populous states. Journal of managed care & specialty pharmacy, 27(3), 354-366.
[4] Ward, Z. J., Bleich, S. N., Long, M. W., & Gortmaker, S. L. (2021). Association of body mass index with health care expenditures in the United States by age and sex. PloS one, 16(3), e0247307.
[5] Wang, Y., Zhao, L., Gao, L., Pan, A., & Xue, H. (2021). Health policy and public health implications of obesity in China. The lancet Diabetes & endocrinology, 9(7), 446-461.
[6] Okunogbe, A., Nugent, R., Spencer, G., Ralston, J., & Wilding, J. (2021). Economic impacts of overweight and obesity: current and future estimates for eight countries. BMJ global health, 6(10), e006351.
[7] Gregg, E. W., Sattar, N., & Ali, M. K. (2016). The changing face of diabetes complications. The lancet Diabetes & endocrinology, 4(6), 537-547.
[8] Bommer, C., Sagalova, V., Heesemann, E., Manne-Goehler, J., Atun, R., Bärnighausen, T., ... & Vollmer, S. (2018). Global economic burden of diabetes in adults: projections from 2015 to 2030. Diabetes care, 41(5), 963-970.
[9] Jayedi, A., Rashidy-Pour, A., Soltani, S., Zargar, M. S., Emadi, A., & Shab-Bidar, S. (2020). Adult weight gain and the risk of cardiovascular disease: a systematic review and dose–response meta-analysis of prospective cohort studies. European journal of clinical nutrition, 74(9), 1263-1275.
[10] Younossi, Z., Anstee, Q. M., Marietti, M., Hardy, T., Henry, L., Eslam, M., ... & Bugianesi, E. (2018). Global burden of NAFLD and NASH: trends, predictions, risk factors and prevention. Nature reviews Gastroenterology & hepatology, 15(1), 11-20.
[11] Basen-Engquist, K., & Chang, M. (2011). Obesity and cancer risk: recent review and evidence. Current oncology reports, 13, 71-76.
[12] Pati, S., Irfan, W., Jameel, A., Ahmed, S., & Shahid, R. K. (2023). Obesity and cancer: A current overview of epidemiology, pathogenesis, outcomes, and management. Cancers, 15(2), 485.
[13] Calle, E. E., & Thun, M. J. (2004). Obesity and cancer. Oncogene, 23(38), 6365-6378.
[14] Fulton, S., Décarie-Spain, L., Fioramonti, X., Guiard, B., & Nakajima, S. (2022). The menace of obesity to depression and anxiety prevalence. Trends in Endocrinology & Metabolism, 33(1), 18-35.
[15] Wolk, R., Shamsuzzaman, A. S., & Somers, V. K. (2003). Obesity, sleep apnea, and hypertension. Hypertension, 42(6), 1067-1074.
[16] Peppard PE, Young T, Palta M, Dempsey J, Skatrud J. Longitudinal study of moderate weight change and sleep-disordered breathing. J Am Med Assoc. 2000;284:3015–21
[17] Fildes A, Charlton J, Rudisill C, Littlejohns P, Prevost AT, Gulliford MC. Probability of an Obese Person Attaining Normal Body Weight: Cohort Study Using Electronic Health Records. Am J Public Health. 2015 Sep;105(9):e54-9.
[18] AP, L. C. S., Fussman, C., & Imes, G. Fast-Food Consumption and Obesity Among Michigan Adults.
[19] Rosenheck, R. (2008). Fast food consumption and increased caloric intake: a systematic review of a trajectory towards weight gain and obesity risk. Obesity reviews, 9(6), 535-547.
[20] Zhang, C., Kaye, J. A., Cai, Z., Wang, Y., Prescott, S. L., & Liberles, S. D. (2021). Area postrema cell types that mediate nausea-associated behaviors. Neuron, 109(3), 461-472.
[21] Williams, E. K., Chang, R. B., Strochlic, D. E., Umans, B. D., Lowell, B. B., & Liberles, S. D. (2016). Sensory neurons that detect stretch and nutrients in the digestive system. Cell, 166(1), 209-221.
[22] O'Neil, P. M., Birkenfeld, A. L., McGowan, B., Mosenzon, O., Pedersen, S. D., Wharton, S., ... & Wilding, J. P. (2018). Efficacy and safety of semaglutide compared with liraglutide and placebo for weight loss in patients with obesity: a randomised, double-blind, placebo and active controlled, dose-ranging, phase 2 trial. The Lancet, 392(10148), 637-649.
[23] Garvey, W. T., Batterham, R. L., Bhatta, M., Buscemi, S., Christensen, L. N., Frias, J. P., ... & STEP 5 Study Group. (2022). Two-year effects of semaglutide in adults with overweight or obesity: the STEP 5 trial. Nature medicine, 28(10), 2083-2091.
[24] Zhang, C., Vincelette, L. K., Reimann, F., & Liberles, S. D. (2022). A brainstem circuit for nausea suppression. Cell reports, 39(11).
[25] Rosenstock, J., Wysham, C., Frías, J. P., Kaneko, S., Lee, C. J., Landó, L. F., ... & Thieu, V. T. (2021). Efficacy and safety of a novel dual GIP and GLP-1 receptor agonist tirzepatide in patients with type 2 diabetes (SURPASS-1): a double-blind, randomised, phase 3 trial. The Lancet, 398(10295), 143-155.
[26] Jastreboff, A. M., Aronne, L. J., Ahmad, N. N., Wharton, S., Connery, L., Alves, B., ... & Stefanski, A. (2022). Tirzepatide once weekly for the treatment of obesity. New England Journal of Medicine, 387(3), 205-216.
[27] Knop, F. K., Aroda, V. R., do Vale, R. D., Holst-Hansen, T., Laursen, P. N., Rosenstock, J., ... & Garvey, W. T. (2023). Oral semaglutide 50 mg taken once per day in adults with overweight or obesity (OASIS 1): a randomised, double-blind, placebo-controlled, phase 3 trial. The Lancet.
[28] Pratt, E., Ma, X., Liu, R., Robins, D., Haupt, A., Coskun, T., ... & Benson, C. (2023). Orforglipron (LY3502970), a novel, oral non‐peptide glucagon‐like peptide‐1 receptor agonist: A Phase 1a, blinded, placebo‐controlled, randomized, single‐and multiple‐ascending‐dose study in healthy participants. Diabetes, Obesity and Metabolism.
[29] Pratt, E., Ma, X., Liu, R., Robins, D., Haupt, A., Coskun, T., ... & Benson, C. (2023). Orforglipron (LY3502970), a novel, oral non‐peptide glucagon‐like peptide‐1 receptor agonist: A Phase 1a, blinded, placebo‐controlled, randomized, single‐and multiple‐ascending‐dose study in healthy participants. Diabetes, Obesity and Metabolism.
[30] LAI-SAN THAM, MELISSA K. THOMAS, CHARLES BENSON, ROSS BRAY, CHENG CAI TANG, CORINA LOGHIN; 106-OR: A First-in-Human Single Ascending Dose Study of Oxyntomodulin Analog LY3305677 in Healthy Subjects. Diabetes 1 June 2021; 70 (Supplement_1): 106–OR
[31] Innovent Biologics Clinical Data Update Mazdutide (IBI362) Higher Dose 9mg Phase 2 Clinical study in obesity, May 11, 2023.
[32] Ji, L., Gao, L., Jiang, H., Yang, J., Yu, L., Wen, J., ... & Qian, L. (2022). Safety and efficacy of a GLP-1 and glucagon receptor dual agonist mazdutide (IBI362) 9 mg and 10 mg in Chinese adults with overweight or obesity: a randomised, placebo-controlled, multiple-ascending-dose phase 1b trial. EClinicalMedicine, 54.
[33] Knerr, P. J., Mowery, S. A., Douros, J. D., Premdjee, B., Hjøllund, K. R., He, Y., ... & Finan, B. (2022). Next generation GLP-1/GIP/glucagon triple agonists normalize body weight in obese mice. Molecular Metabolism, 63, 101533.
[34] Bossart, Martin, et al. "Effects on weight loss and glycemic control with SAR441255, a potent unimolecular peptide GLP-1/GIP/GCG receptor triagonist." Cell metabolism 34.1 (2022): 59-74.
[35] Choi, I. Y., Lee, J. S., Kim, J. K., Park, Y. J., Jung, S. Y., Kim, Y. H., & Kwon, S. C. (2017). Potent body weight loss and efficacy in a NASH animal model by a novel long-acting GLP-1/Glucagon/GIP triple-agonist (HM15211). American Diabetes Association’s 77th Scientific Session.
[36] M. Abdelmalek, J. Choi, Y. Kim, K. Seo, M. Hompesch, S. Baek, HM15211, a novel GLP-1/GIP/glucagon triple-receptor co-agonist significantly reduces liver fat and body weight in obese subjects with non-alcoholic fatty liver disease: a phase 1b/2a, multi-center, randomized, placebo-controlled trial, J. Hepatol. 73 (2020) S124,
[37] Coskun, T., Urva, S., Roell, W. C., Qu, H., Loghin, C., Moyers, J. S., ... & Milicevic, Z. (2022). LY3437943, a novel triple glucagon, GIP, and GLP-1 receptor agonist for glycemic control and weight loss: From discovery to clinical proof of concept. Cell Metabolism, 34(9), 1234-1247.
[38] Rosenstock, J., Frias, J., Jastreboff, A. M., Du, Y., Lou, J., Gurbuz, S., ... & Coskun, T. (2023). Retatrutide, a GIP, GLP-1 and glucagon receptor agonist, for people with type 2 diabetes: a randomised, double-blind, placebo and active-controlled, parallel-group, phase 2 trial conducted in the USA. The Lancet.
[39] Nestor, J. J., Zhang, X., Jaw‐Tsai, S., Parkes, D. G., & Becker, C. K. (2021). Design and characterization of a surfactant‐conjugated, long‐acting, balanced GLP‐1/glucagon receptor dual agonist. Peptide Science, 113(5), e24221.
[40]https://ir.altimmune.com/news-releases/news-release-details/altimmune-announces-positive-results-week-24-interim-analysis
[41] Heymsfield, S. B., Coleman, L. A., Miller, R., Rooks, D. S., Laurent, D., Petricoul, O., ... & Roubenoff, R. (2021). Effect of bimagrumab vs placebo on body fat mass among adults with type 2 diabetes and obesity: a phase 2 randomized clinical trial. JAMA network open, 4(1), e2033457-e2033457.
[42] Morley, J. E., & Flood, J. F. (1991). Amylin decreases food intake in mice. Peptides, 12(4), 865-869.
[43] Frias, J. P., Deenadayalan, S., Erichsen, L., Knop, F. K., Lingvay, I., Macura, S., ... & Davies, M. (2023). Efficacy and safety of co-administered once-weekly cagrilintide 2· 4 mg with once-weekly semaglutide 2· 4 mg in type 2 diabetes: a multicentre, randomised, double-blind, active-controlled, phase 2 trial. The Lancet.
[44] https://doi.org/10.2337/db23-51-OR
[45] Foley, J. E., & Jordan, J. (2010). Weight neutrality with the DPP-4 inhibitor, vildagliptin: mechanistic basis and clinical experience. Vascular Health and Risk Management, 541-548.
[46] Enebo, L. B., Berthelsen, K. K., Kankam, M., Lund, M. T., Rubino, D. M., Satylganova, A., & Lau, D. C. (2021). Safety, tolerability, pharmacokinetics, and pharmacodynamics of concomitant administration of multiple doses of cagrilintide with semaglutide 2· 4 mg for weight management: a randomised, controlled, phase 1b trial. The Lancet, 397(10286), 1736-1748.
[47] Steinert, R. E., Poller, B., Castelli, M. C., Drewe, J., & Beglinger, C. (2010). Oral administration of glucagon-like peptide 1 or peptide YY 3-36 affects food intake in healthy male subjects. The American journal of clinical nutrition, 92(4), 810-817.
[48] Connolly, H. M., Crary, J. L., McGoon, M. D., Hensrud, D. D., Edwards, B. S., Edwards, W. D., & Schaff, H. V. (1997). Valvular heart disease associated with fenfluramine–phentermine. New England Journal of Medicine, 337(9), 581-588.
[49] Sharretts, J., Galescu, O., Gomatam, S., Andraca-Carrera, E., Hampp, C., & Yanoff, L. (2020). Cancer risk associated with lorcaserin—the FDA’s review of the CAMELLIA-TIMI 61 trial. New England Journal of Medicine, 383(11), 1000-1002.
[50] The mechanism of action for Rimonabant was cannabinoid receptor CB1 antagonism, in other words, blocking the cannabinoid receptor (the one that weed, for example, binds to and activates in order to make you all happy). Seems like a predictable outcome that it would make you quite sad. There is a company currently called Agentix that is investigating a new CB1 receptor antagonist as an antiobesity drug and, very unfortunately, it’s OTC and only has a market cap of 3 million or else this entire article would be about why everyone should short it.
[51] Vickers, S. P., Clifton, P. G., Dourish, C. T., & Tecott, L. H. (1999). Reduced satiating effect of d-fenfluramine in serotonin 5-HT2C receptor mutant mice. Psychopharmacology, 143, 309-314.
[52] Xu, Y., Jones, J. E., Lauzon, D. A., Anderson, J. G., Balthasar, N., Heisler, L. K., ... & Elmquist, J. K. (2010). A serotonin and melanocortin circuit mediates D-fenfluramine anorexia. Journal of Neuroscience, 30(44), 14630-14634.
[53] The mechanism of action for Rimonabant was cannabinoid receptor CB1 antagonism, in other words, blocking the cannabinoid receptor (the one that weed, for example, binds to and activates in order to make you all happy). Seems like a predictable outcome that it would make you quite sad. There is a company currently called Agentix that is investigating a new CB1 receptor antagonist as an antiobesity drug and, very unfortunately, it’s OTC and only has a market cap of 3 million or else this entire article would be about why everyone should short it.
[54] Beiroa, D., Imbernon, M., Gallego, R., Senra, A., Herranz, D., Villarroya, F., ... & Nogueiras, R. (2014). GLP-1 agonism stimulates brown adipose tissue thermogenesis and browning through hypothalamic AMPK. Diabetes, 63(10), 3346-3358.
[55] Vangoitsenhoven, R., Mathieu, C., & Van der Schueren, B. (2012). GLP1 and cancer: friend or foe?. Endocrine Related Cancer, 19(5), F77.
[56] Petersen, K. M., Bøgevig, S., Riis, T., Andersson, N. W., Dalhoff, K. P., Holst, J. J., ... & Christensen, M. B. (2020). High‐Dose Glucagon Has Hemodynamic Effects Regardless of Cardiac Beta‐Adrenoceptor Blockade: A Randomized Clinical Trial. Journal of the American Heart Association, 9(21), e016828.
[57] Ambery, P. D., Klammt, S., Posch, M. G., Petrone, M., Pu, W., Rondinone, C., ... & Hirshberg, B. (2018). MEDI0382, a GLP‐1/glucagon receptor dual agonist, meets safety and tolerability endpoints in a single‐dose, healthy‐subject, randomized, phase 1 study. British journal of clinical pharmacology, 84(10), 2325-2335.
[58] https://doi.org/10.2337/db23-95-LB
[59] https://doi.org/10.2337/db23-763-P
[60] https://doi.org/10.2337/db23-760-P
[61] https://doi.org/10.2337/db23-754-P
[62] https://www.biopharmcatalyst.com/company/GPCR/news/138739
[63] https://doi.org/10.2337/db22-335-OR
[64] Pioneering next-generation peptide therapeutics, Zealand Pharma, June 2023
[65] https://doi.org/10.2337/db23-92-LB
[66] Dutton, G. R., Bodell, L. P., Smith, A. R., & Joiner, T. E. (2013). Examination of the relationship between obesity and suicidal ideation. International journal of obesity, 37(9), 1282-1286
[67] Carpenter, K. M., Hasin, D. S., Allison, D. B., & Faith, M. S. (2000). Relationships between obesity and DSM-IV major depressive disorder, suicide ideation, and suicide attempts: results from a general population study. American journal of public health, 90(2), 251.
EDIT: Added after original publication for release to free subscribers









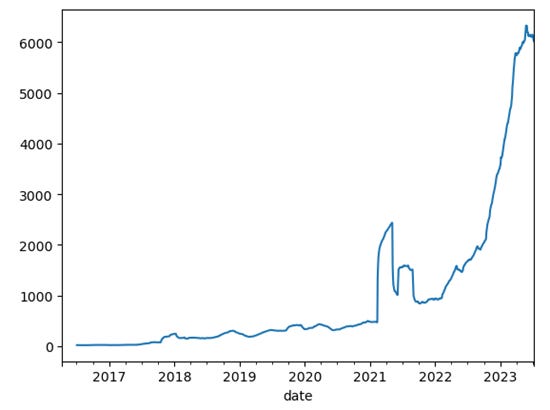
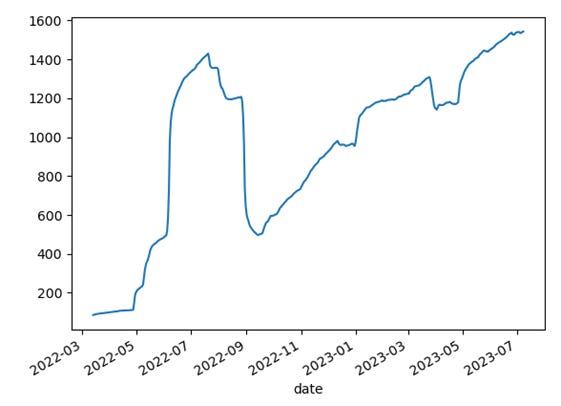




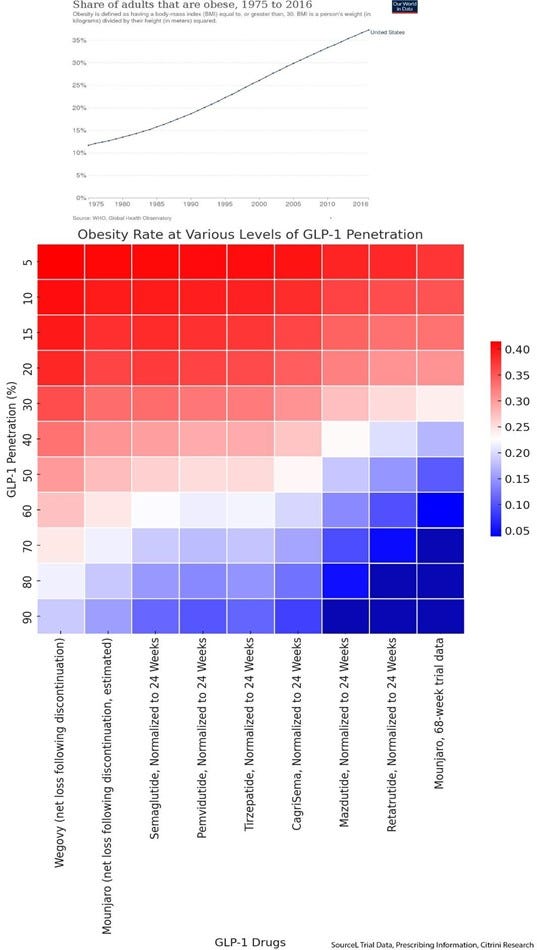

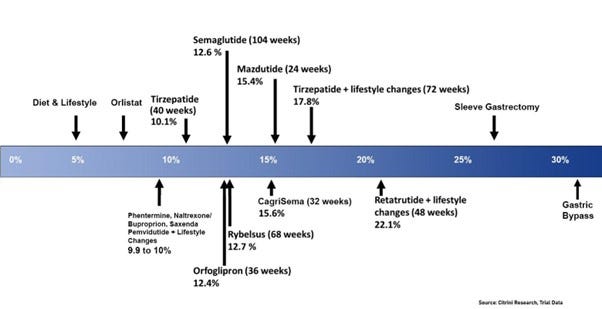
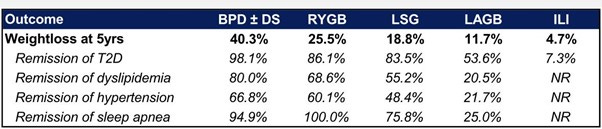





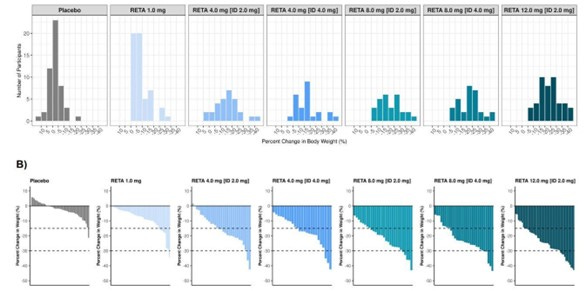
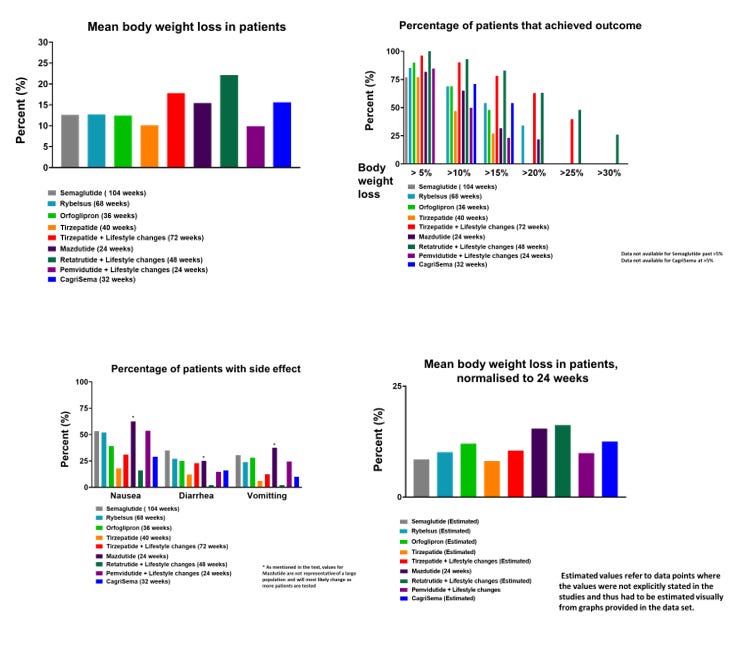

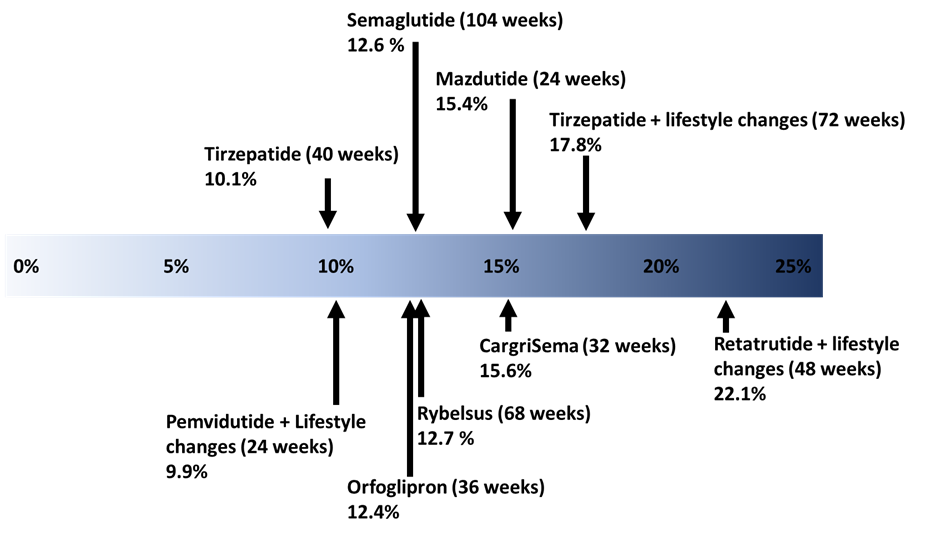

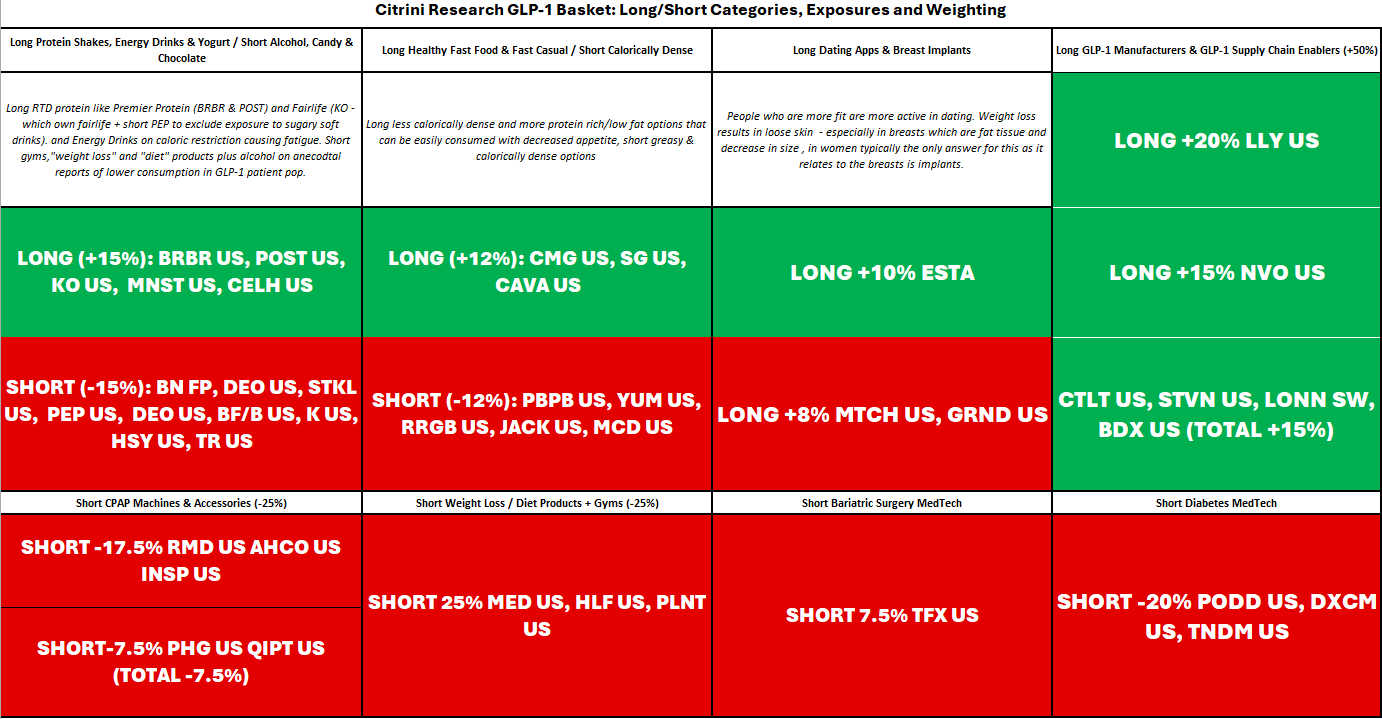




Would be great to see this change in the society!
Before going to the beach for their TikTok I guess people will have to do something for their loose skins. After losing weight this much that is bound to happen. Any ticker for plastic surgery, skincare? That should also fit in your thesis on luxury goods/services(?)
Great thesis and writeup. Planet Fitness (PLNT) is an excellent short as in addition to what you mentioned Edwin Dorsey of TheBearCave substack highlighted a number of issues with it